
Our Product
NX210c – An innovative peptide targeting a broad range of neurodegenerative diseases
NX210c is a short, synthetic peptide inspired by SCO-spondin, a multifunctional glycoprotein essential in neurogenesis. It targets blood-brain barrier restoration, supports neuronal protection, and enhances intercellular communication. Its mechanism of action may have therapeutic relevance across multiple CNS disorders.

Origin of NX210c
The scientific journey began decades ago with research on neurogenesis during embryonic development at the University of Clermont-Ferrand (France), focusing on SCO-spondin, a multifunctional glycoprotein secreted by the subcommissural organ.
The Subcommissural Organ (SCO)
The human subcommissural organ (SCO), an ependymal derivative of the diencephalic roof, shows its highest development between the 3rd and 4th months of fetal life, followed by a gradual regression during the second half of pregnancy and a further decline after birth. Thus, in 6-year-old children, only rudiments of SCO parenchyma can be found.
SCO-spondin
SCO-spondin: a multifunctional glycoprotein produced by the subcommissural organ is essential for neurogenesis during embryogenesis.
Intellectual Property
NX210c is protected by 7 fully owned patent families including a composition of matter already granted in EU/USA/JP.
NX210c has also been granted “Orphan Drug Designation” (ODD) by the FDA and the EMA for ALS indication.
A multi-target mechanism of action
Restore the blood-brain barrier
Restores the blood-brain barrier to prevent toxic infiltration into the brain
Neuroprotection
Protects neurons against glutamate excitotoxicity and promotes cellular survival
Enhance Neurotransmission
Improves synaptic communication between neurons
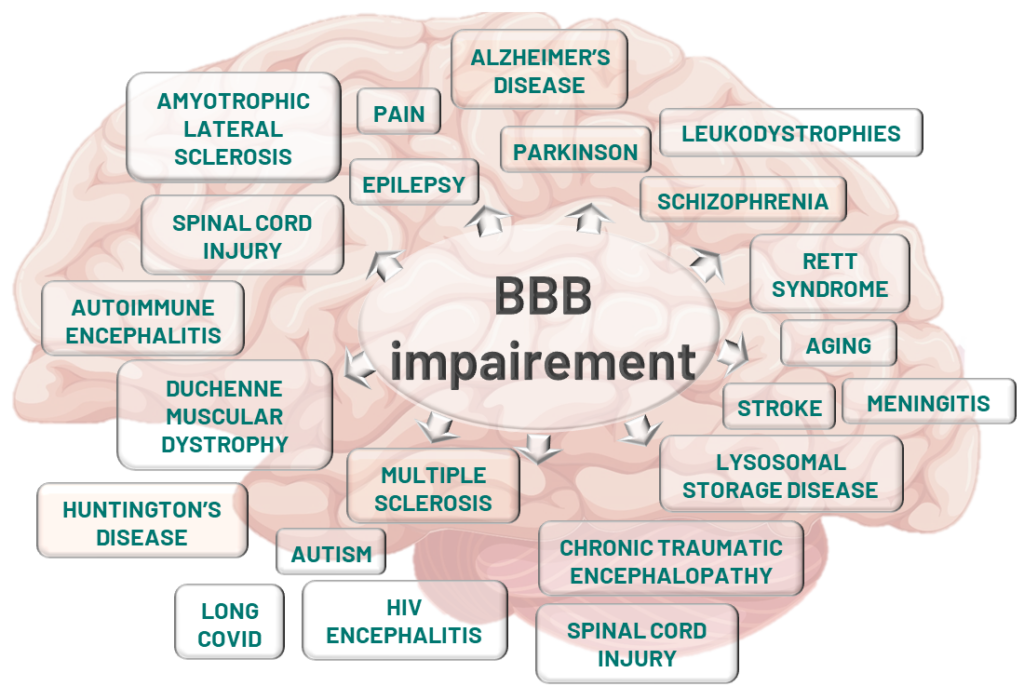
The BBB: a key player in CNS disease progression
Axoltis is developing NX210c, a short amino acid peptide with unique properties that directly targets the restoration of the blood-brain barrier (BBB), a key player in the onset and progression of numerous central nervous system (CNS) diseases.
Scientific Consensus
There is now consensus in the scientific community regarding the causal link between BBB disruption and the development of CNS disorders – including neurodegenerative diseases, stroke, and traumatic brain injury.
A New Therapeutic Strategy
While past research often focused on increasing BBB permeability to allow therapeutic agents to pass through, recent literature highlights the critical importance of preserving BBB integrity.
Tight Junction Proteins
Specifically, reduced expression of tight junction proteins such as claudin-5 has been strongly correlated with increased BBB permeability and the onset of diseases like Alzheimer’s, Parkinson’s, Amyotrophic Lateral Sclerosis, Huntington’s disease, multiple sclerosis, and schizophrenia—both in patients and in animal models. Considering this, repairing and protecting the BBB has emerged as a promising new therapeutic strategy.
SEALS — Phase 2 Clinical Trial in ALS
The SEALS study is a Phase 2, multicenter, randomized, double-blind, placebo-controlled clinical trial evaluating NX210c in adult patients with amyotrophic lateral sclerosis (ALS). Coordinated by Dr. Bernard at the ALS Reference Center of Hospices Civils de Lyon, the study assesses the efficacy, safety, tolerability, and pharmacokinetics of repeated intravenous administrations of NX210c. Patients are stratified based on baseline serum Neurofilament Light (NfL) levels to support robust clinical evaluation. The study is registered on ClinicalTrials.gov (NCT06365216). Recruitment for this clinical trial is now closed. No additional patients can be enrolled.
To learn more about reusing clinical trial data, click here.
Therapeutic potential across CNS disorders
NX210c’s mechanism of action extends its therapeutic potential beyond ALS, our first indication. By restoring blood-brain barrier integrity, protecting neurons and enhancing the neurotransmission, NX210c may have relevance across a broad spectrum of CNS disorders.

*Quantitative System Pharmacology